39 electron configuration and orbital notation worksheet answers
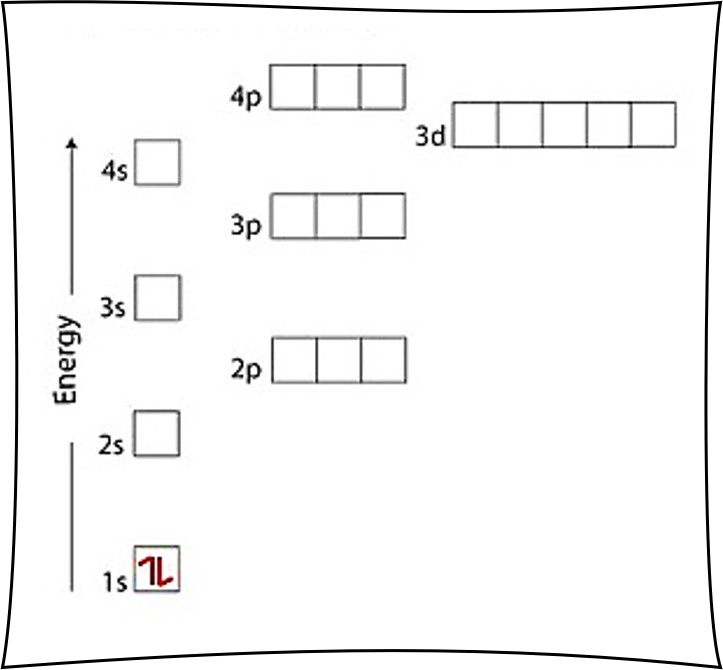
6. Complete the electron configuration portion of the table on your student answer sheet. ORBITAL NOTATION Orbital notation is a drawing of the electron configuration. It is very useful in determining electron pairing and thus predicting oxidation numbers. The orbital notation for sulfur would be represented as follows:
Created Date: 10/5/2015 12:33:05 PM
Orbital notation: 1s222s262p6 23s 103p 4s 3d10 4p6 5s 4d 5p6 6s2 4f14 5d4 Orbital notation + Arrows: Noble gas notation: [Xe] 6s2 4f14 5d4 g) Tellurium Orbital notation:21s 2s2 2p6 3s 3p 4s2 3d10 4p6 5s2 4d10 5p4 Orbital notation + Arrows: Noble gas notation:[Kr]5s24d105p4 2. Write the ground state electron configuration for neutral atom Iodine ...
Electron configuration and orbital notation worksheet answers
Electron configuration worksheet answer key. Use the Aufbau principle to fill the orbitals with electrons for elements 1-23. Since there is 1 electron in the box labeled 1s we say the H electron configuration in orbital notation is 1s 1. Download Chemquest 30 Intro To Moles Answer Key nocReadCom PDF for free.
-Determining electron configuration from number of electrons -Determining the identity of an element from its electron configuration -Completing orbital diagrams using arrows to represent electrons . Title: Electron Configuration Author: Brian T. Walker Created Date:
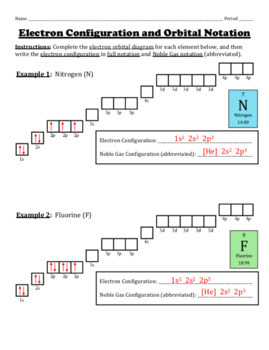
Electron Configurations and Orbital Diagrams KEY ... Write the electron configuration (full, and in core notation): 1. scandium ... Answer the following questions: 1. Describe the two differences between a 2p x orbital and a 3p y orbital. The 2px orbital lies on the x-axis. The 3py orbital lies on the y-axis and is larger than the 2px orbital.
Electron configuration and orbital notation worksheet answers.
Electron Configurations and Periodic Trends 1. Rt has three extra electrons 11 02 2 12. Orbital Diagrams Doc Chemistry Classroom Teaching Chemistry Electron Configuration Just fancy it by voting.Electron configuration worksheet 3 answer key. The electron configurations in this worksheet assume that lanthanum la is the first element in the 4f block and that actinium […]
calcium has 20 electrons. No matter what the atom is, the orbital structure is the same. In class we will learn how to use the periodic table to remember the orbital structure, and then write it using the shorthand notation of electron configurations. Some things to remember: • Each orbital can contain 0, 1, or 2 electrons (and no more!).
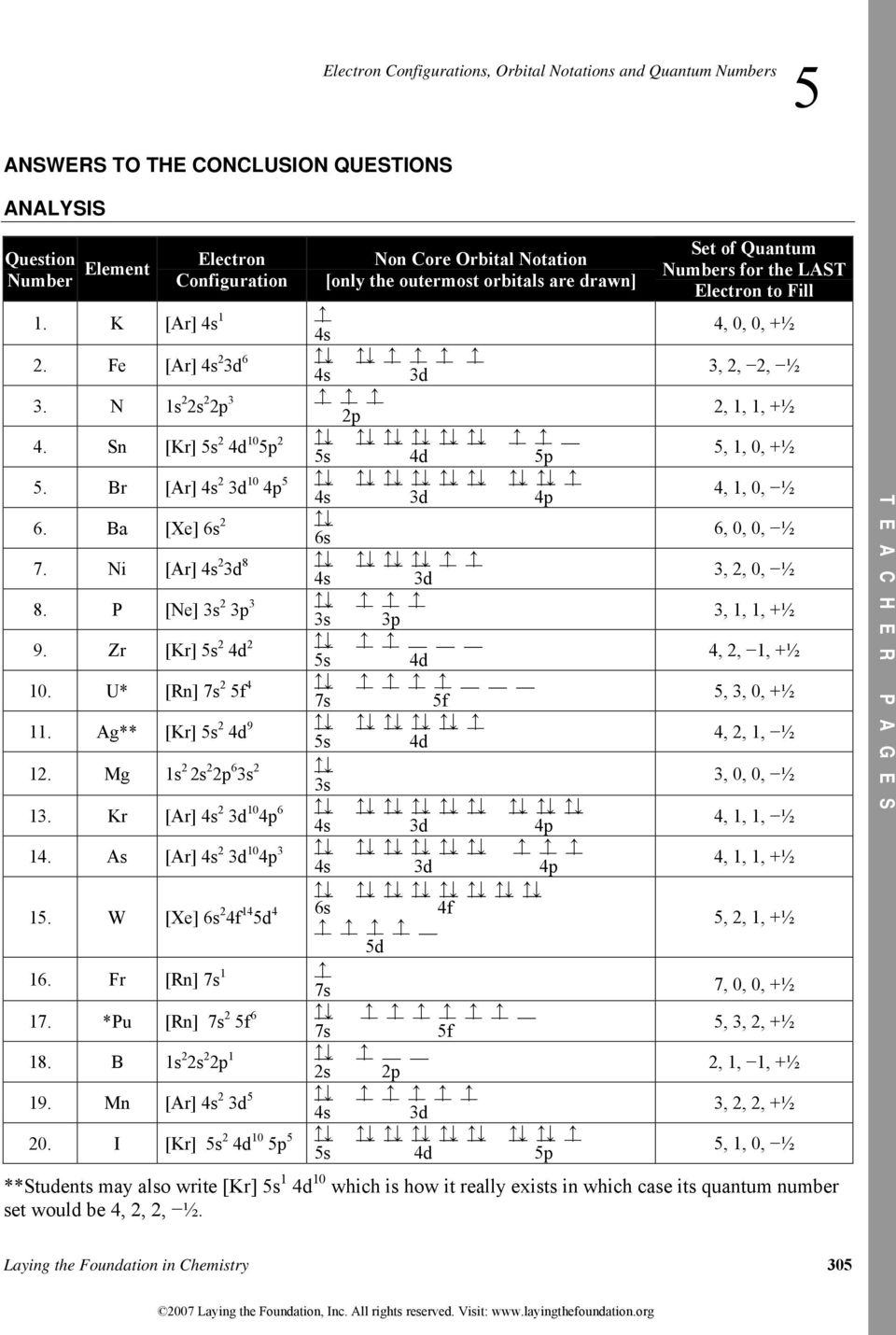
Electron Configuration, Orbital Notation, Electron Dot and Valence Electrons Electron Dot Be valence electrons Name Element and atomic # 26 He 20 25 33 Be Electron Configuration Is22s22p63s23p2 s22s22p63s23p6 3d64s2 15 L 2 s 12 Orbital Notation Is 2s U 3p 3p L IL U 110 U Is 2s LLLLL 11 3d Is 2s 3d 11 Is U . Created Date:
38. The ground state electron configuration of Fe is _____. A. 2 1s 2s 2 3s 3p6 3d6 B. 2 1s 2 2s 2p6 3s2 3p6 4s 3d6 C. 2 1s2 2s 2p6 3s2 3p6 4s2 D. 2 1s 2 2s 2p6 3s 3p6 4s2 4d6 39. The ground state electron configuration of Ga is _____. A. 2 1s 2s2 3s2 3p6 3d10 4s2 4p1 B. 6 1s 2 2s 2p 3s2 3p6 4s2 4d10 4p1 C. 2 1s2 2s 2p6 3s2 3p6 4s2 3d10 4p1
Electrons always fill up the lowest energy orbital first. Write the electron configuration and orbital notations for the following atoms and ions. Displaying top 8 worksheets found for orbital notation. The order of filling in electrons in the subshells is 1s 2s 2p 3s 3p 4s s subshells have only 1 orbital 2 electrons per orbital 2 electrons max.
The electron configuration orbital diagram worksheet answers can be found at the bottom of the lesson The 2 8 8 18 rule is a very simplistic view of electron configuration and doesnt give the full picture when it comes to electron configuration.
Electron Orbitals: Electron Configuration Orbital Diagram Worksheet Answers (The electron configuration orbital diagram worksheet answers can be found at the bottom of the lesson.) The 2, 8, 8, 18 rule is a very simplistic view of electron configuration and doesn't give the full picture when it comes to electron configuration.
View Homework Help - Orbital Notation and Electron Configuration Answer Key.pdf from CHEMISTRY 1 at Plano East Sr H S. Orbital Notation and Electron Configurations Answer Key Write the electron
Since there is 1 electron in the box labeled 1s, we say the H electron configuration in orbital notation is 1s 1. The orbital notation can also be interpreted as quantum numbers, where the principal quantum number n is the energy level (1 before the s), the azimuthal quantum number corresponds to the letter s, and the spin quantum number is +1/2.
Electron Configurations. The content that follows is the substance of General Chemistry Lecture 26. In this lecture we continue the discussion of Quantum Numbers and their use in Electron Configurations as well as the relationship of electron configuration to the periodic properties of the elements.
Ad The most comprehensive library of free printable worksheets digital games for kids. Electron configuration worksheet answer key. Configurations of ions present a. Rt has three extra electrons 11 02 2 12. The electron configurations in this worksheet assume that lanthanum la is the first element in the 4f block and that actinium ac is the first.
O If you think of regular electron configuration as giving specific directions, Noble Gas configuration gives much more general directions O For example, if someone is trying to get to Lake Ridge from Fort Worth, you can give them turn by turn directions.
All groups and messages ... ...
Electron configuration worksheet 2 answer key. The idea is to draw an arrow for each electron so in this case we just have one arrow to draw. The 6 key answers for the electron configuration chem worksheet 5 are. Electron Configurations - Solutions Note. Is 2s lectron Is 4s on 2s a o o gurations or ome Orbital filling elected ements Electron ...
Electron Configurations Worksheet Write the complete ground state electron configurations and orbital notations for the following: # of e-Element ... In the space below, write the orbital notation (arrows) of the following elements: 11) manganese _____ 12) silver _____ ...
—While electron configurations provide some information about the location of electrons in an atom, orbital notations provide all four quantum numbers and thus, the "complete address" of electrons. Energy levels, sublevels, # of orbitals, and spin of electrons are all provided and shown with orbital notations.
Using the periodic table answer the following questions: 16) Which Groups have an s-orbital as the last orbital? 1 and 2. 17) Which Groups have a p-orbital as the last orbital? 13 - 18. 18) Which Groups have a d-orbital as the last orbital? 3 - 12. Which section of the table is left? What is trend of f-orbital elements? Lanthanide and actinide
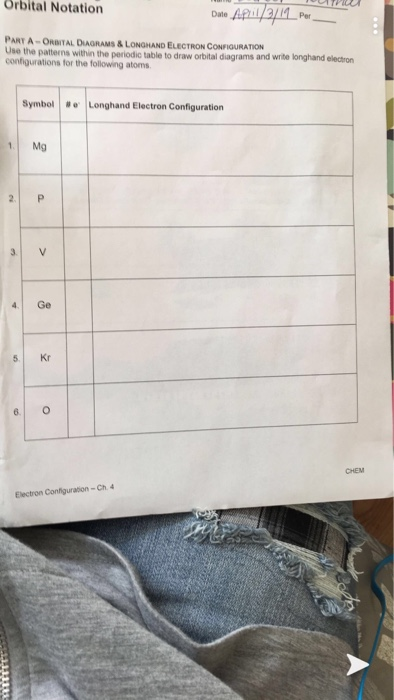
Stability & Electron Configuration – Ch. 4 CHEM PART B – SHORTHAND ELECTRON CONFIGURATION Use the patterns within the periodic table to write the longhand electron configuration notation for the following elements. Symbol # e- Longhand Electron Configuration Notation 7. S 1s2 2s2 2p6 3s2 3p4 8. Pb 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 ...
Practice: Write electron distributions and do the orbital notation for the following: 1. P : 2. Ca: Only do the electron distributions for the following: 1. Co: 2. Eu: 3. Tc: orbitals Arows quantum elctron n l m s n enrgy l orbital ua nt mbers energy sublevel orbital
View Electron Configurations and Orbital Notations Worksheet.docx from CHEM. 4766 at Cooper City High School. Name: Allison Gross_ Date: 10/28/20_ Class period: 8_ Unit: Electron
Dec 07, 2021 · The 6 key answers for the electron configuration chem worksheet 5 are. The sum of the superscripts should equal the total number of electrons. Select the option to save image as. Since there is 1 electron in the box labeled 1s we say the H electron configuration in orbital notation is 1s 1.
Electron Configuration Worksheet This worksheet provides extra practice for writing electron configurations. The easiest and most reliable technique for writing electron configurations is to use the periodic table as your guide. Details of using the periodic table as a guide for determining electron configurations can be found on the CH301 website.
If an orbital contains 2 electrons, they must have opposite “spin.” Hund’s rule: One electron must occupy each orbital in a sublevel before a second electron occupies an orbital. Part B – Rules of Electron Configurations. Which of the following “rules” is being violated in each electron configuration below? Explain your answer for each.
This is a collection of 6 (as of October 2019) of my first-year chemistry worksheets covering electrons, electron and ion configurations, orbital notation, noble-gas shorthand, and basic electromagnetic spectrum. I run a very high-energy, interactive lab/class. These activities are easy to grade but









![6) cobalt [Ar] 4s 2 3d 7](https://s2.studylib.net/store/data/009918562_1-1950b3428f2f6bf78209e86f923b4abf.png)



















0 Response to "39 electron configuration and orbital notation worksheet answers"
Post a Comment