39 redox reactions practice worksheet
Instructions on balancing chemical equations: Enter an equation of a chemical reaction and click 'Balance'. The answer will appear below; Always use the upper case for the first character in the element name and the lower case for the second character.
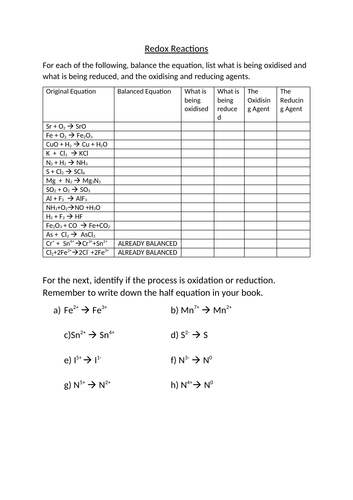
Practice Problems: Redox Reactions. Determine the oxidation number of the elements in each of the following compounds: a. H 2 CO 3 b. N 2 c. Zn(OH) 4 2-d. NO 2-e. LiH f. Fe 3 O 4 Hint; Identify the species being oxidized and reduced in each of the following reactions: a. Cr + + Sn 4+ Cr 3+ + Sn 2+ b. 3 Hg 2+ + 2 Fe (s) 3 Hg 2 + 2 Fe 3+ c. 2 As (s) + 3 Cl 2 (g) 2 AsCl 3 Hint; …
Balancing Redox Reactions Worksheet 1 Balance each redox reaction in . acid. solution. Mn 2+ + BiO3 -Æ MnO4 -+ Bi 3+ MnO4 -+ S2O3 2- Æ S4O6 2- + Mn 2+ ClO3 - + Cl - Æ Cl2 + ClO2 . P + Cu 2+ Æ Cu + H2PO4 -PH3 + I2 Æ H3PO2 -+ I -NO2 Æ NO3 -+ NO . Basic Solutions . MnO4 -+ C2O4 2- Æ MnO2 + CO2 . ClO2 Æ ClO2 -+ ClO3 -

Redox reactions practice worksheet
Apr 17, 2013 · Balancing REDOX Reactions: Learn and Practice Reduction-Oxidation reactions (or REDOX reactions) occur when the chemical species involved in the reactions gain and lose electrons. Oxidation and reduction occur simultaneously in order to conserve charge. We can “see” these changes if we assign oxidation numbers to the reactants and products.
Chapter 5: Introduction to Redox Chemistry 5.3: Types of Chemical Reactions ... Many combustion reactions occur with a hydrocarbon, a compound made up solely of carbon and hydrogen. The products of the combustion of hydrocarbons are always carbon dioxide and water. Many hydrocarbons are used as fuel because their combustion releases very large amount of …
08.05.2013 · page 5 Redox practice worksheet. 41. Base your answer(s) to the following question(s) on the diagram below. The diagram shows a voltaic cell with copper and aluminum electrodes immediately after the external circuit is completed. As this voltaic cell operates, the mass of the Al(s) electrode decreases. Explain, in terms of particles, why this decrease in …
Redox reactions practice worksheet.
Some chemical reactions do not fit nicely into one of the four categories of chemical reactions. This different class of reactions is called REDOX reactions, which will be discussed, in later chapters. Ex) C2H5OH + 3O2 ( 2CO2 + 3H2O. Chapter 8 Reaction Prediction. For each of the following equations: Identify the type of reaction.
Practice Problems: Redox Reactions (Answer Key) Determine the oxidation number of the elements in each of the following compounds: a. H 2 CO 3 H: +1, O: -2, C: +4 b. N 2 N: 0 c. Zn(OH) 4 2-Zn: 2+, H: +1, O: -2 d. NO 2-N: +3, O: -2 e. LiH Li: +1, H: -1 f. Fe 3 O 4 Fe: +8/3, O: -2; Identify the species being oxidized and reduced in each of the following reactions: a. Cr + + …
Jun 28, 2010 · Chapter 20 Worksheet: Redox ANSWERS I. Determine what is oxidized and what is reduced in each reaction. Identify the oxidizing agent and the reducing agent, also. 1. 2Sr + O2 2SrO Sr 0 to Sr2+; oxidized/reducing agent O0 to O2-; reduced/ox. ag. 2. 2Li + S Li2S Li 0 to Li1+; oxidized/red. ag. S0 to S2-; reduced/ox. ag. 3.
Worksheet # 5 Balancing Redox Reactions in Acid and Basic Solution Balance each half reaction in basic solution. 4. Cr 2O 7 2 - → Cr3+ 5. NO → NO 3-6. SO 4 2- → SO 2 7. MnO 2 → Mn 2O 3 Balance each redox reaction in acid solution using the half reaction method. 8. H 2O 2 + Cr 2O 7 2- → O 2 + Cr 3+ 9. TeO 3 2-+ N 2O 4 → Te + NO 3-10. ReO 4-+ IO-→ IO 3-+ Re 11. …
25.08.2021 · A redox reaction is a chemical reaction that involves a transfer of electrons and changes in oxidation number. Learn about redox reactions, and identify oxidizing and reducing agents, and explore ...
WS # 4 Balancing Redox Reactions . Balance each of the following half-cell reactions. (In each case assume that the reaction takes place in an ACIDIC solution.) Also, state whether the reaction is oxidation or reduction. 1.
16.08.2021 · Redox Reactions & Electron Carriers in Cellular Respiration: Definitions and Examples Quiz Instructions: Choose an answer and click 'Next'. You will receive your score and answers at the end.
Balancing Redox Equations WorkSheet Oxidation Number Method for Balancing Redox Equations 1. 2. 3. 4. 5. 6. 7. Assign oxidation numbers to all elements and identify those that are oxidized and reduced. If only one element is both oxidized and reduced (disproportionation), write it down twice (then recombine it after the equation is balanced).
































0 Response to "39 redox reactions practice worksheet"
Post a Comment